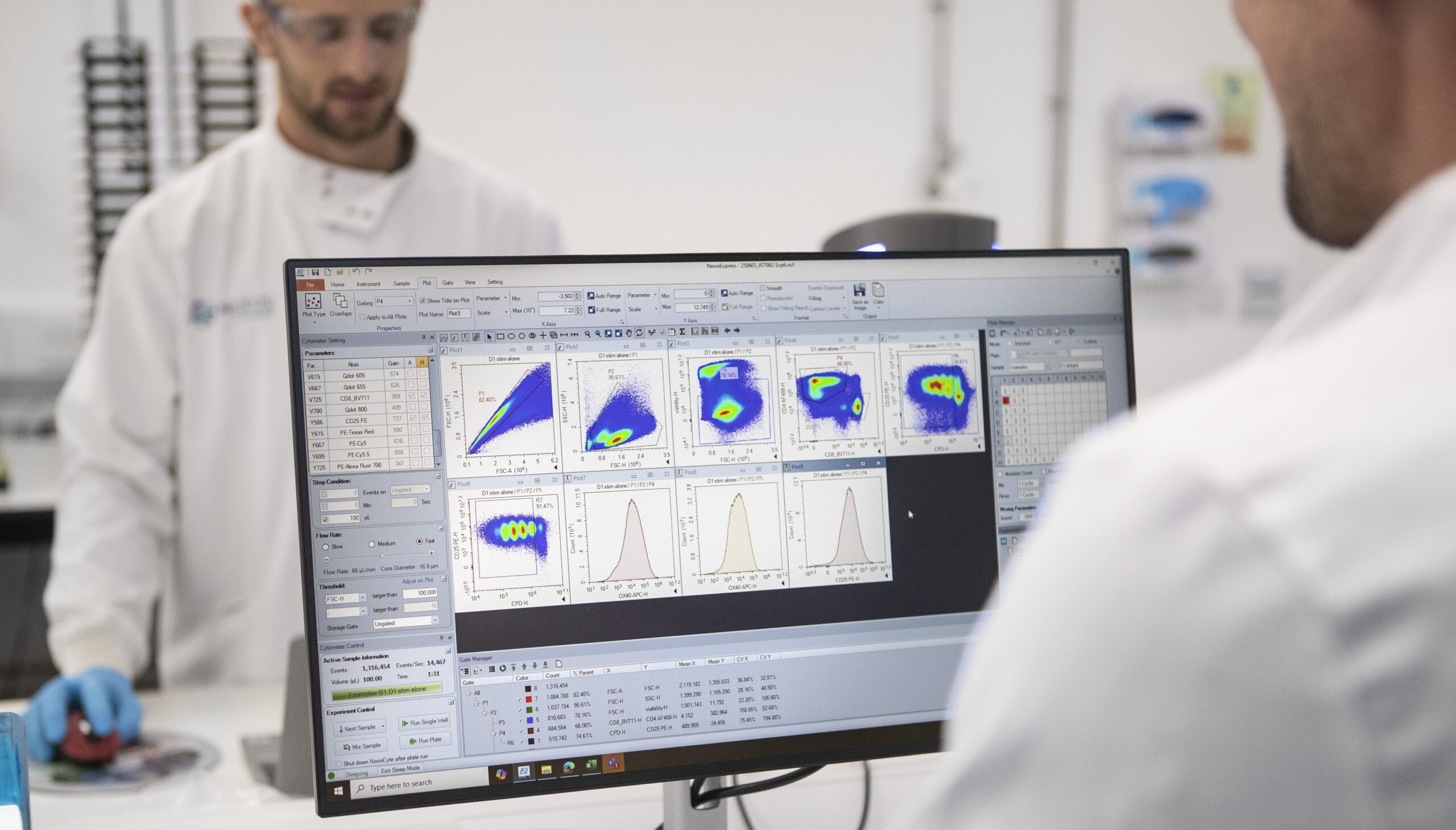
We validate targets in primary human cells and established cell lines, assessing target expression profiles through flow cytometry or qPCR under various disease relevant stimulation conditions. Thereafter, tailored in-vitro functional assays, specific to the cell type and target, are used to confirm the impact of target inhibition or engagement. This is achieved using gene knockout functional assays or direct pathway inhibition with known modulators.