CD4+ T cell
Immune-Oncology Assays
- Polyclonal
- MLR
- Antigen specificity
- nTreg suppression assay
- iTreg polarisation assay
Autoimmunity Assays
- Th1/2/17 polarisation
- Th17 function
- Tr1/iTreg polarisation
- nTreg suppression
- Tfh, naïve
- CM and EM phenotype/function
- Polyclonal T cell proliferation
- Cytokine release
Inflammation Assays
- Cytokine release
Assays
ELISpot is a highly sensitive method to test immune modulators, novel vaccine candidates or de-risk immunogenicity testing in an antigen-specific CD4 and/or CD8 T cell assay.
| Condition | Aim |
| No Stimulation | Negative control |
| PMA | Positive control |
| CERI (CMV, EBV, RSV, Influenza) | MHC-I restricted peptide pool to evaluate modulation of CD8+ T-cell memory response |
| CPI (CMV, Parainfluenza, Influenza) | Positive protein antigens to evaluate modulation of CD4+ T-cell memory response |
| CEF (CMV, EBV, Influenza) | MHC-I restricted peptide pool to evaluate modulation of CD8+ T-cell memory response |
| Cyclosporin A (CsA) | Inhibition of immune response |
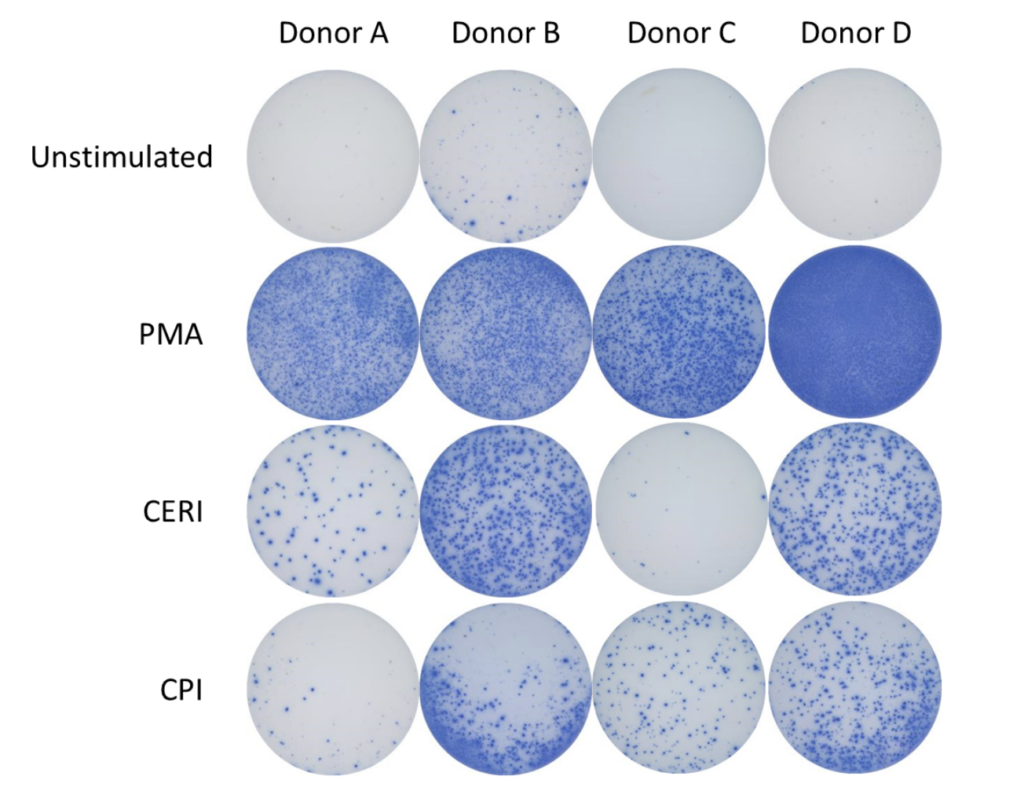
Figure 1: The ELISpot visual image results showing four donors under four conditions: Unstimulated (negative control), stimulated with PMA (positive control), stimulated with CERI, and stimulated with CPI antigens. Each purple spot represents one IFNγ producing cell.
Spot Forming Units (SPU) for IFN-γ per 100,000 PBMC from CERI, CPI and CEF antigens for three donors
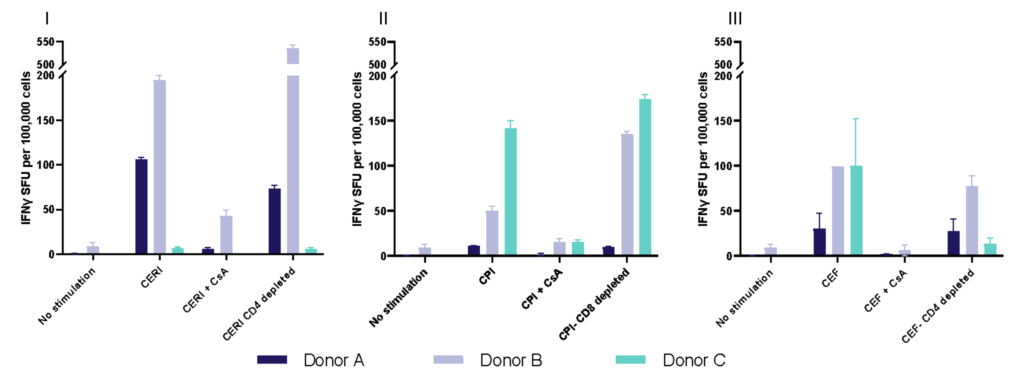
Figure 2: T cell responses from three donors using stimuli, CERI (I), CPI (II) and CEF (III). Expressed as spot forming units per 100,000 donor PBMC. Depletion of CD4 or CD8 T cells demonstrates specificity of response to peptide pools. Each value represents the mean of triplicate wells ±SEM.
T helper 17 (Th17) cells, are a subset of CD4 T cells distinguished by their production of IL-17. While crucial for protecting the body against extracellular pathogens, Th17 cells have also been implicated in autoimmune disease. Discovering therapies that target either the differentiation or effector function of Th17 cells could therefore prove beneficial for the treatment of some autoimmune diseases.
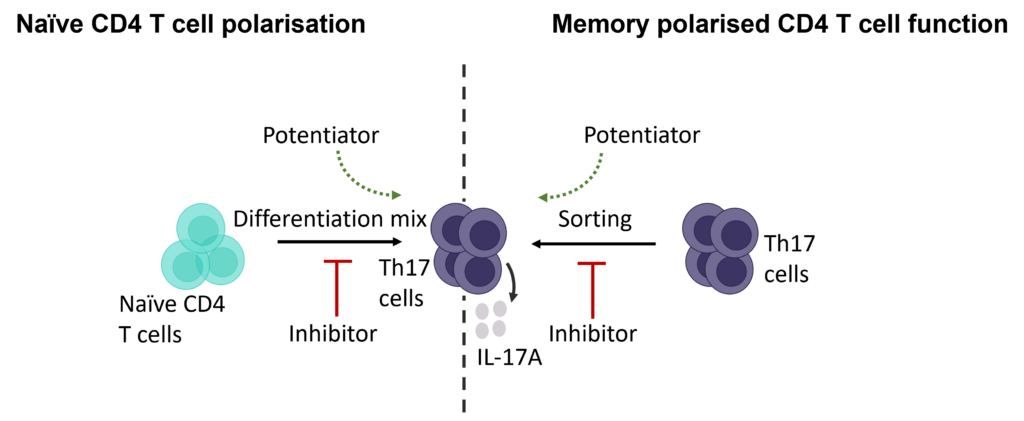
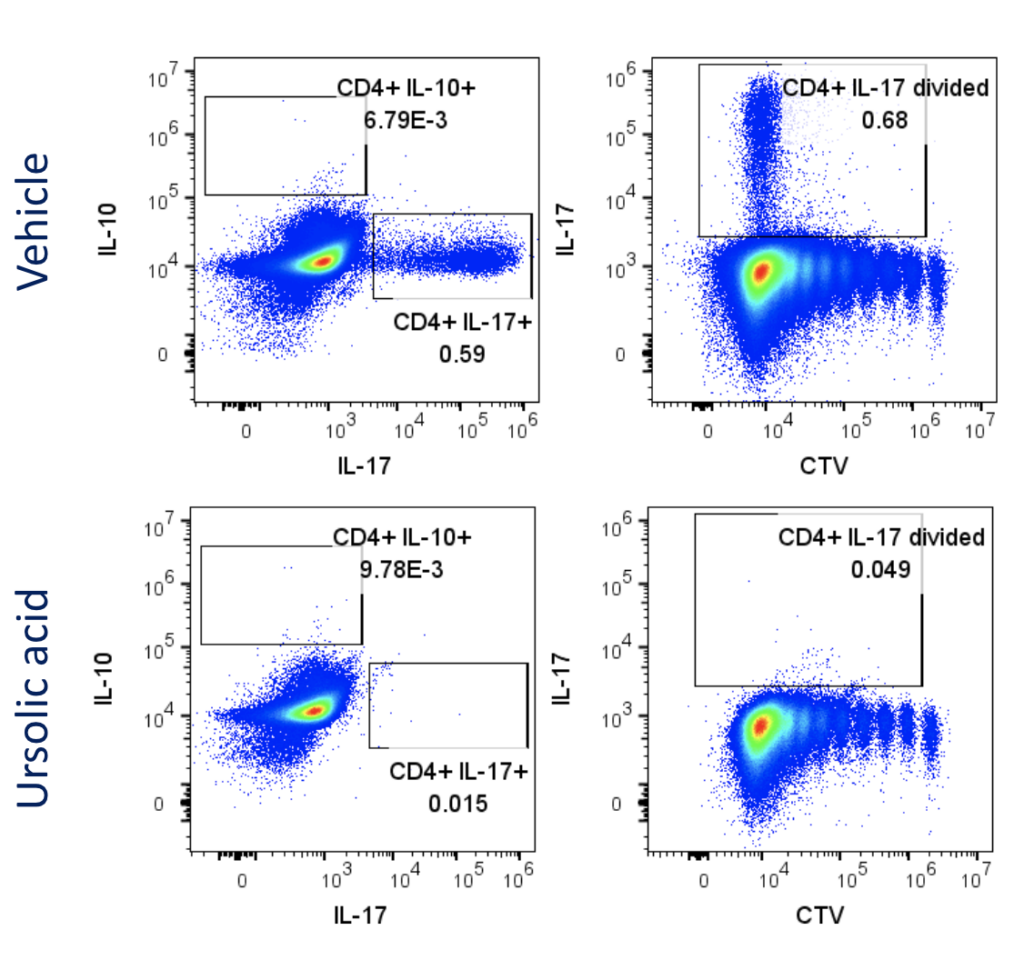
Evaluation of therapeutic modulation of Th17 differentiation
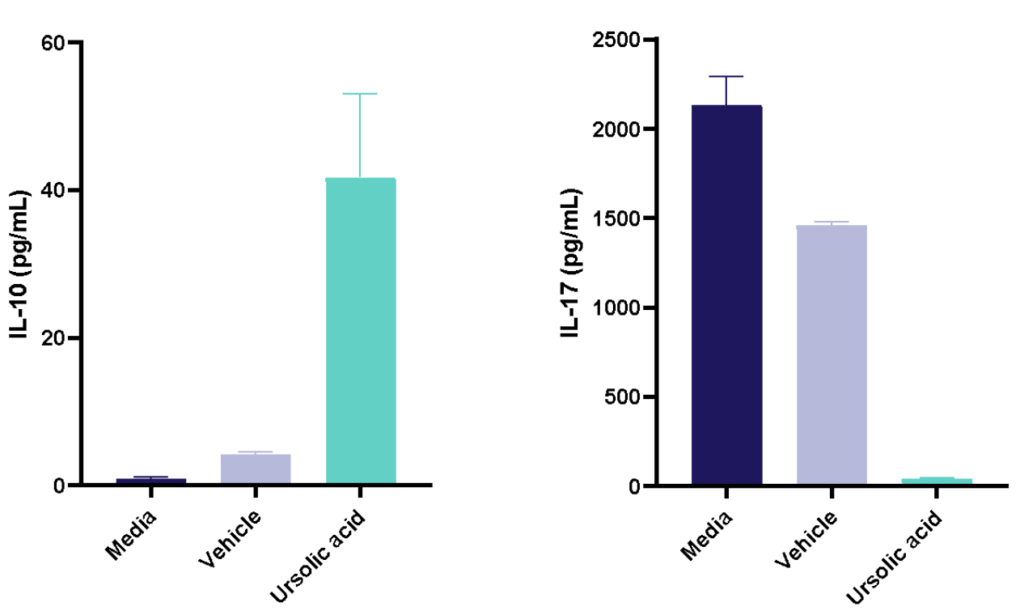
Figure 1: Polarisation of Th17 cells. Naïve CD4 cells were cultured under Th17 polarising conditions for 12 days in the presence or absence of Ursolic Acid. CD4 T cells were assessed for proliferation by CTV dilution; intracellular cytokine staining (ICS) of IL-17 and IL-10 by flow cytometry. On Day 12, Supernatants were collected and evaluated for IL-17 and IL-10 levels by MSD. RoRγT inhibitor Ursolic Acid showed selective inhibition of IL-17 production by intracellular cytokine staining (ICS) and MSD.
Evaluation of therapeutic modulation of Th17 effector function
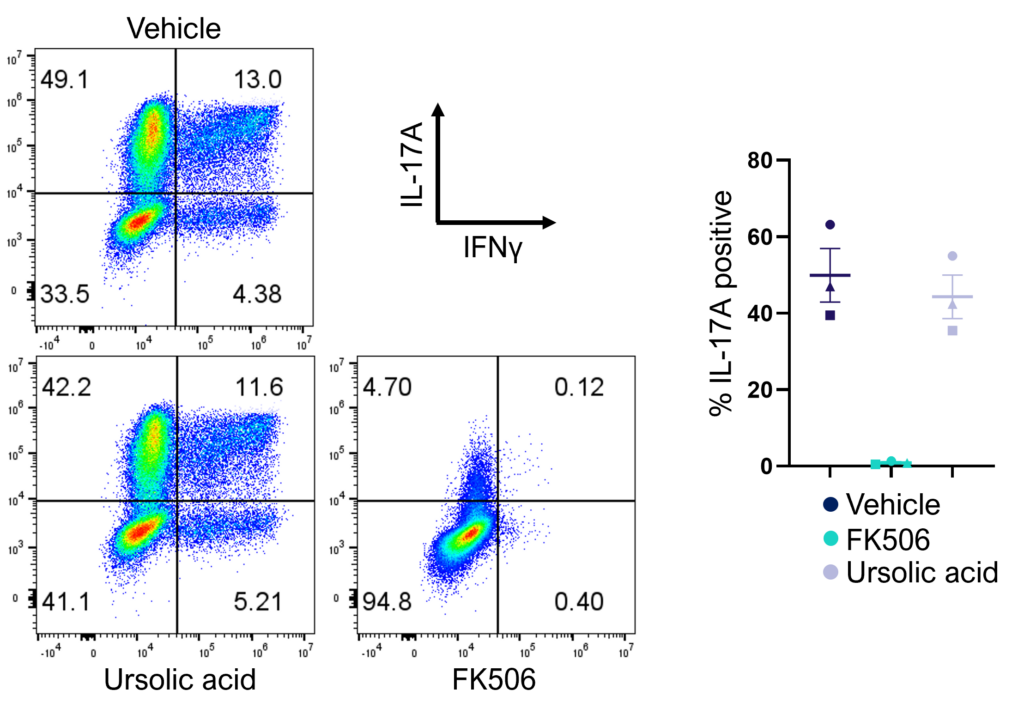
Figure 2: Inhibition of Th17 cell effector function. Magnetically sorted memory Th17 cells were polyclonally stimulated under Th17 conditions in the presence or absence of ursolic acid or FK506 for 5 days. A percentage of Th17 cells are polyfunctional (IL- 17A+IFNγ+) and refractory to inhibition by ursolic acid (an inhibitor of Th17 differentiation) but not FK506.
For many the humble "workhorse" mixed lymphocyte reaction (MLR) is a key screening assay for immuno-therapeutics. 1-way (DC and T cells) and 2-way MLRs (PBMC) provide expression of key ligands on human immune cells for therapeutic modulation.
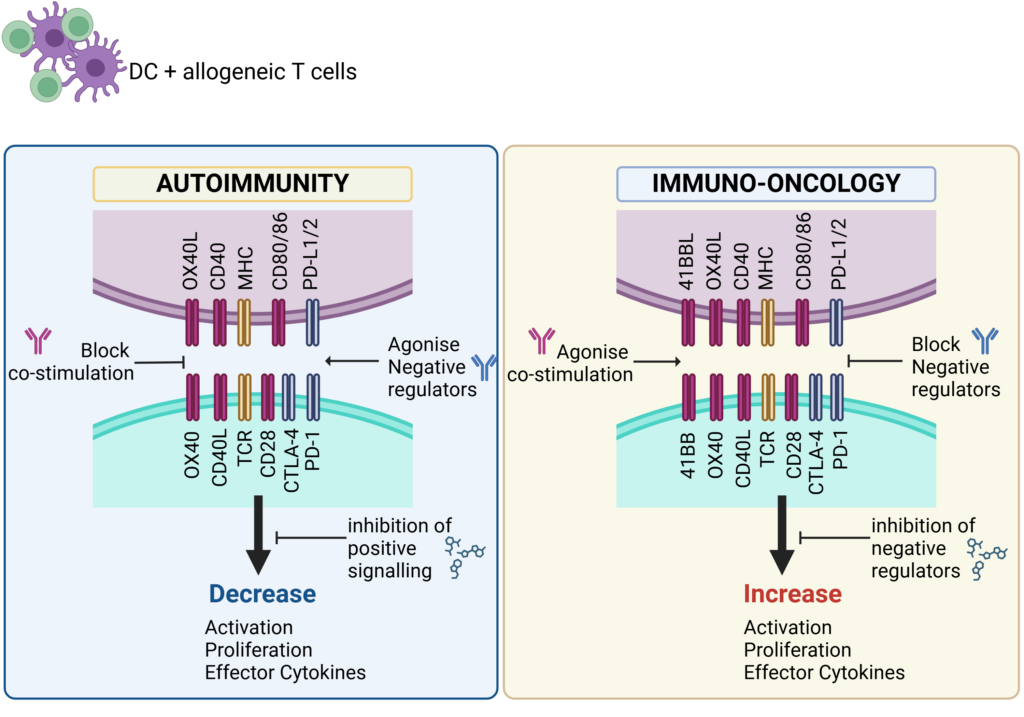
Schematic showing key therapeutic targets that are blocked (antagonise) or engaged (agonise) to control immune responses in Autoimmunity or boost immune responses for Immuno-Oncology.
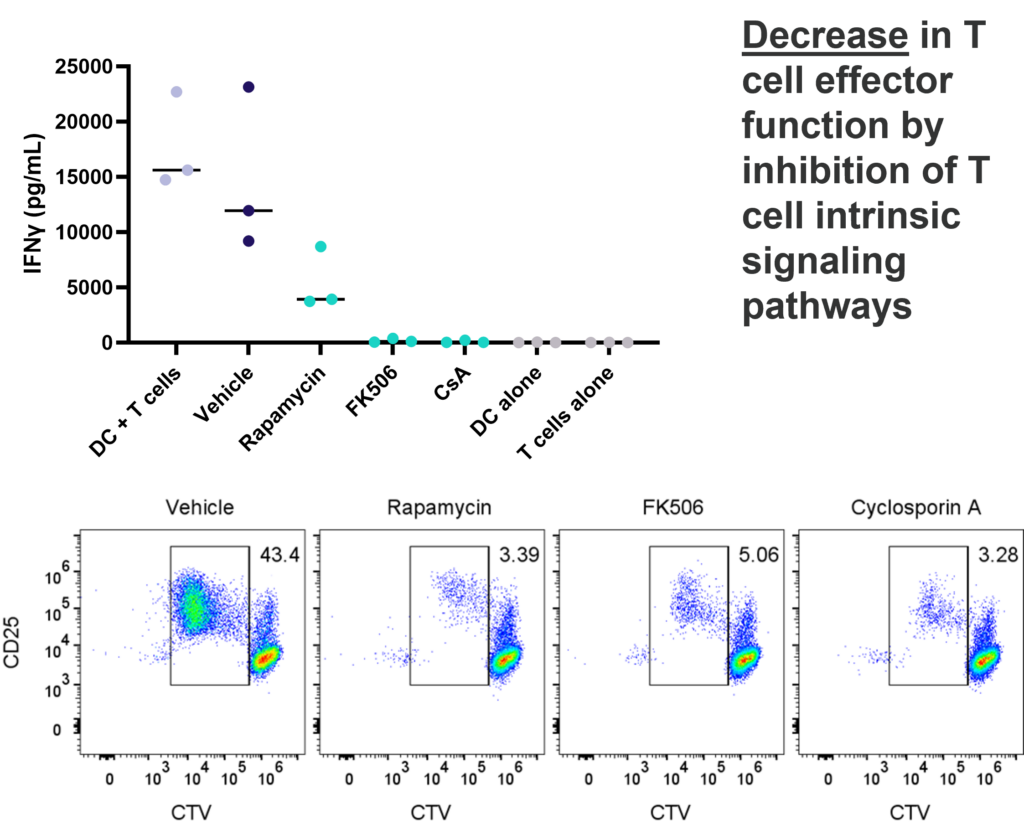

1-way or 2-way MLR. One way MLR of CTV labelled T cells in response to allogenic DC, matured with LPS. T cell proliferation and IFNγ production is inhibited by Rapamycin, FK506 and Cyclosporin A, in contrast, PD-1 blockade (Pembrolizumab) increases T cell IFNγ production, but does not enhance T cell proliferation.
This assay provides a relevant human system to test novel therapeutics that block or engage key co-stimulatory or inhibitory ligands expressed on immune cells.
Natural Tregs (nTregs) are a thymus-derived subset of CD4+ T cells found in peripheral blood, capable of suppressing immune responses to maintain tolerance and homeostasis. Suppression assays model Treg function by measuring their ability to inhibit effector T cell proliferation, activation, and cytokine production. Altering Treg suppressive function can have promising therapeutic potential, both in the context of autoimmunity and immuno-oncology.
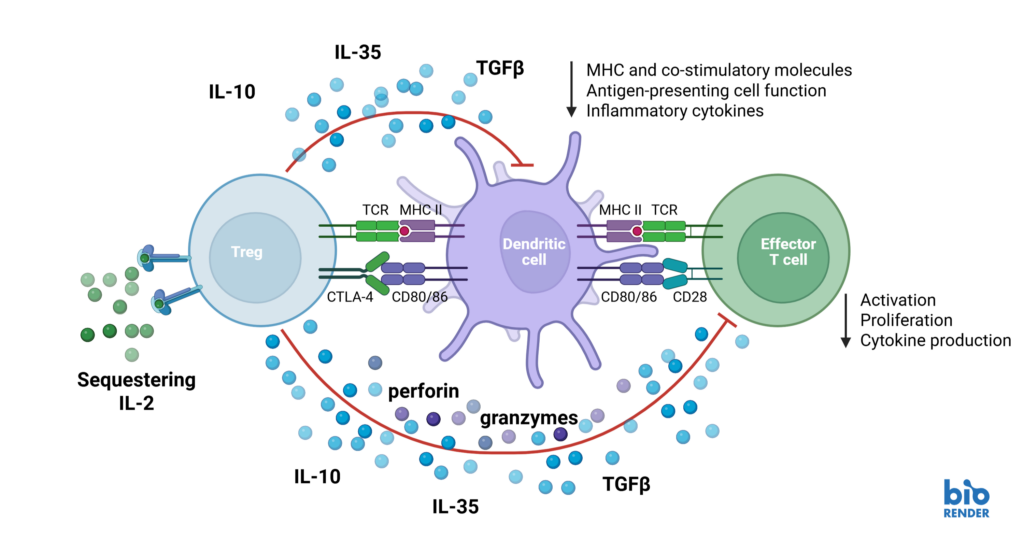
Schematic showing mechanisms by which regulatory T cells suppress effector T cell responses.
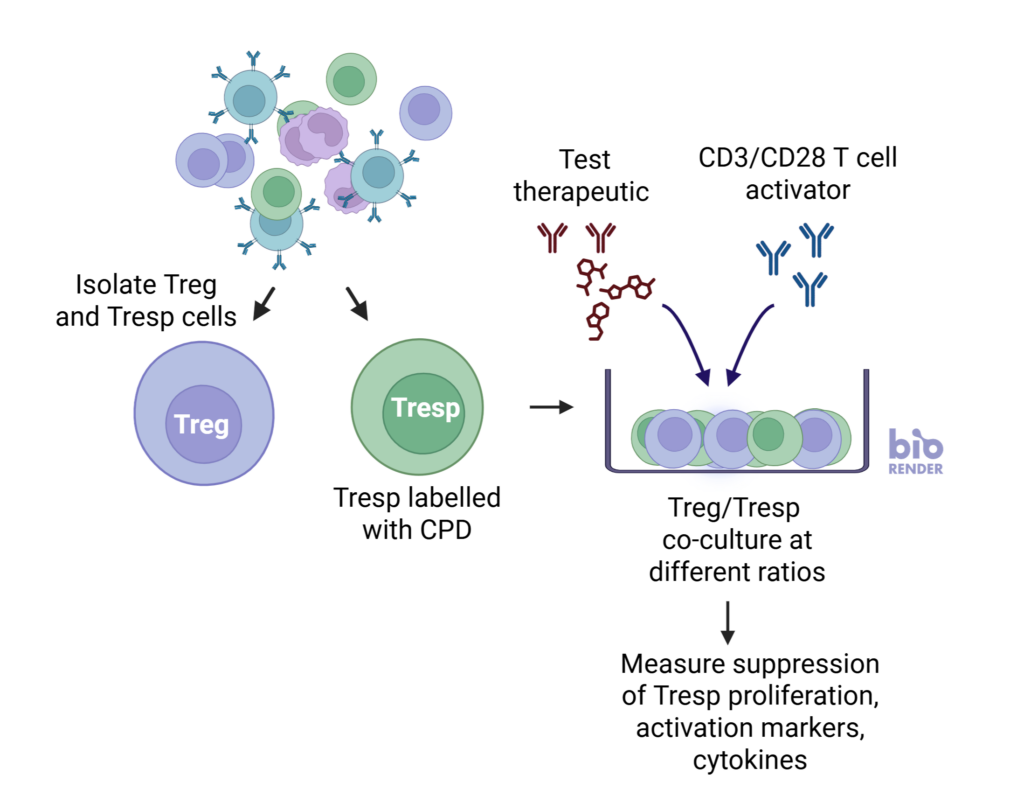
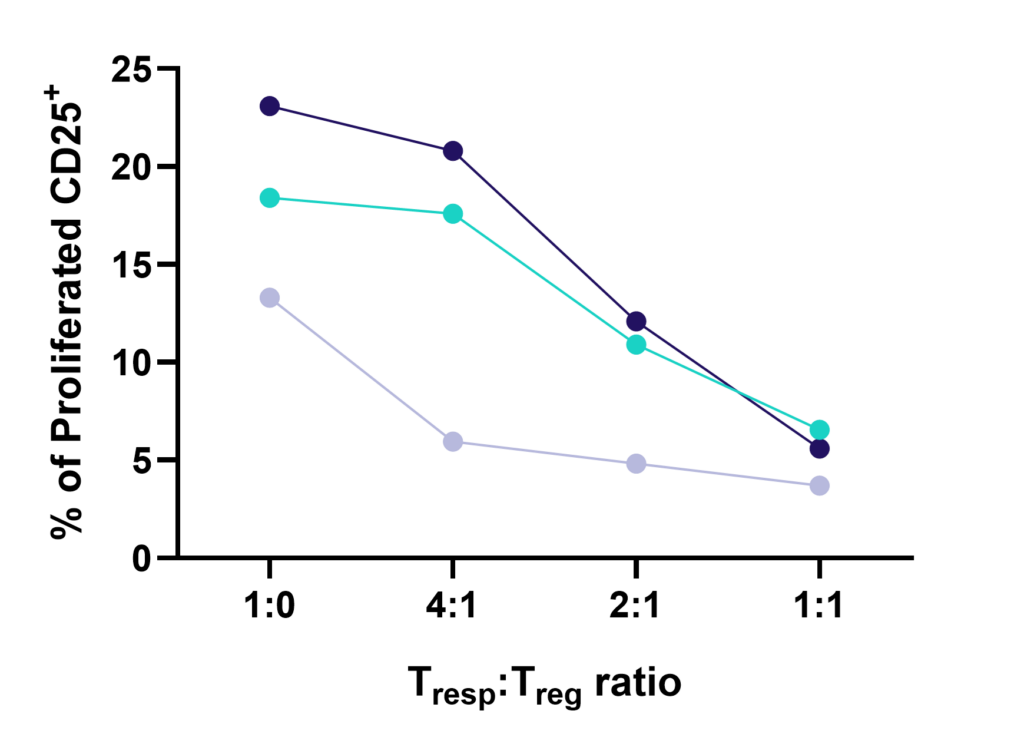
nTreg cells suppress the proliferation of Tresp cells in a ratio-dependent manner
nTregs were co-cultured with stimulated CD3 T cells at three Tresp:Treg ratios. Graphs show increased suppression of Tresponder proliferation in the presence of increasing number of Tregs. Data shown from three healthy donors.
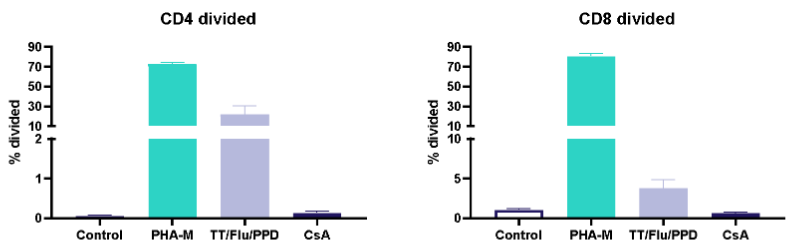
I. T cell response to Tetanus Toxoid, Influenza and PPD antigens
II. Dose response to Influenza antigen
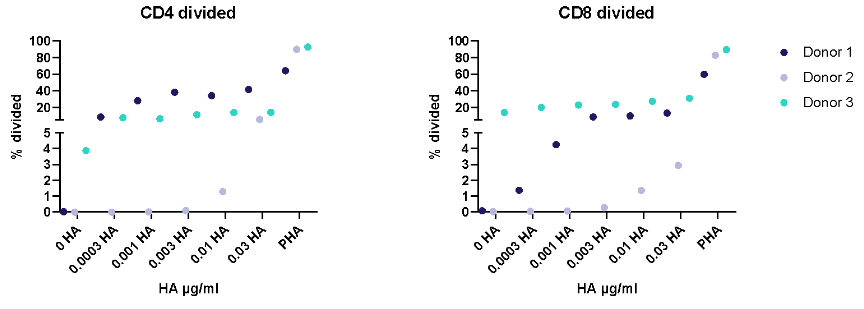
Antigen specific T cells response to a recall antigens. (I) Healthy donors PBMC were stimulated with PHA-M or triple antigen cocktail (Tetanus Toxoid, Influenza and PPD). Cyclosporin was used as a reference treatment. (II) Dose response to Influenza antigen. CD4 and CD8 T cells proliferation was measured by flow cytometry using CTV dilutions.

Drug Discovery Tool
Find the right immune assay for your therapeutic area, modality and target using this interactive Drug Discovery Tool.
