Macrophage
Immune-Oncology Assays
- ADCP
- MDSC
- M1/M2
- TAM
- Efferocytosis
- Phagocytosis
- Cytokine Release Assay (CRA)-predictive toxicity
Inflammation Assays
- M1/M2 polarisation and function
- Cytokine Release Assay (CRA)-predictive toxicity
Assays
Monocyte derived macrophages (moMΦ) are differentiated from monocytes isolated from healthy blood donors followed by polarisation into a range of phenotypes M1/ M2a-d and TAM phenotypes using the stimuli described below. Readouts include flow cytometric analysis of surface markers associated with different subsets, production of cytokines and chemokines and ability to phagocytose. Therapeutics can be added at the monocyte to macrophage differentiation phase or polarisation phase to prevent or drive polarisation into a distinct macrophage phenotype.
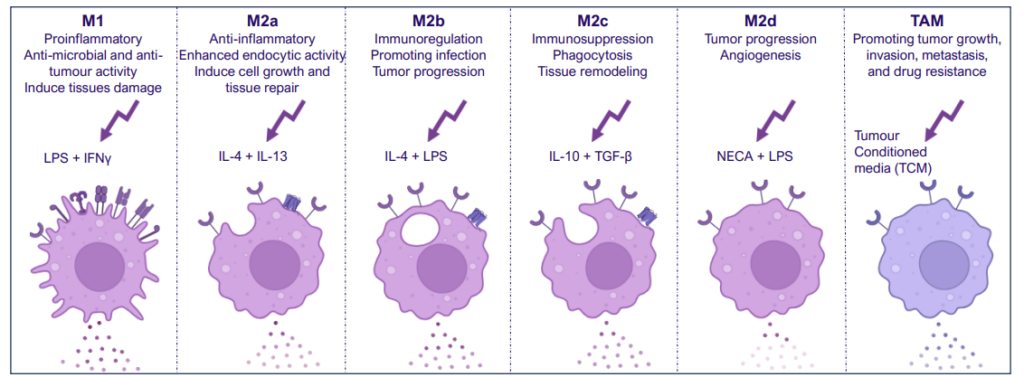
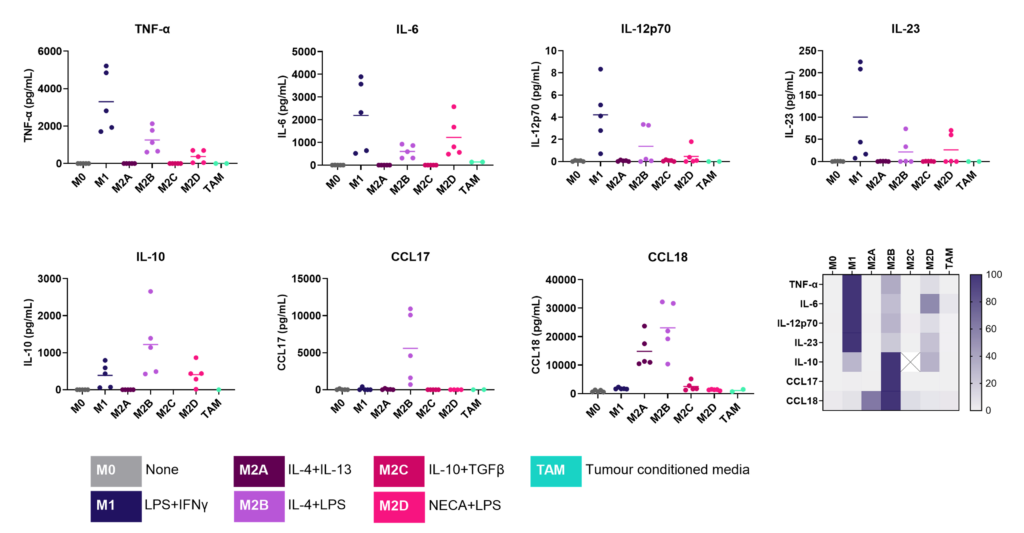
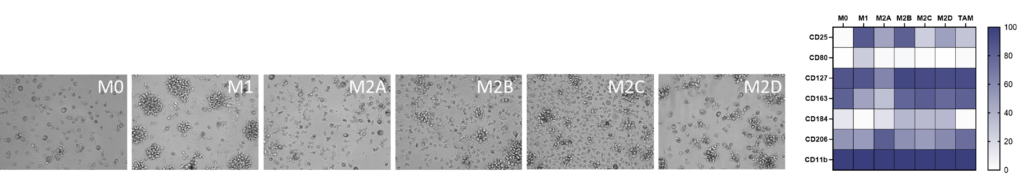
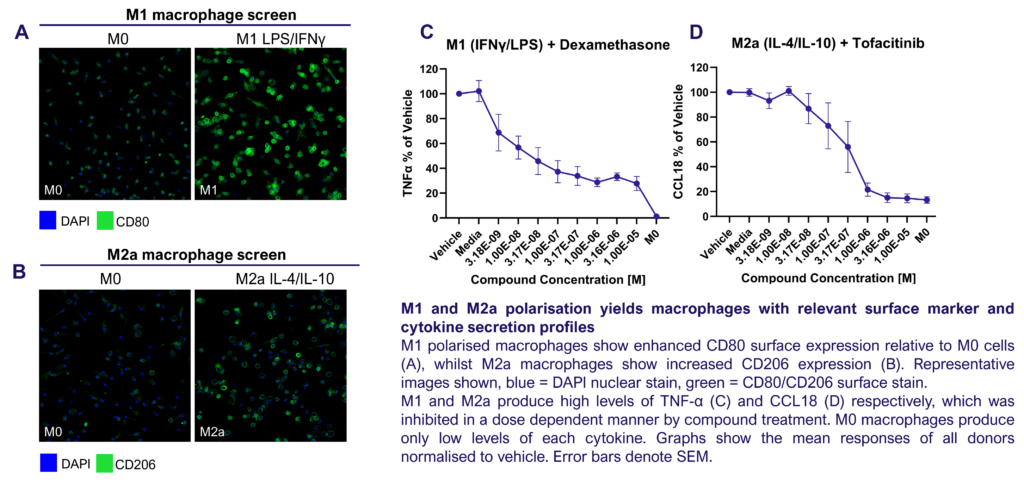
We present a medium-throughput human macrophage assay to screen novel therapeutics that modulate pro-inflammatory M1 (IFNγ/LPS) and anti-inflammatory M2 (IL-4/IL-10) macrophage subsets. Dexamethasone and tofacitinib are used as reference drugs for benchmarking.
Dysregulated macrophage polarisation exacerbates various diseases. Therapeutics that target macrophage re-polarisation could modify the disease state by shifting macrophages to a beneficial phenotype. For example, promoting an inflammatory M1 phenotype could benefit the tumor microenvironment, while fostering an anti-inflammatory phenotype could help in autoimmune and inflammatory diseases.
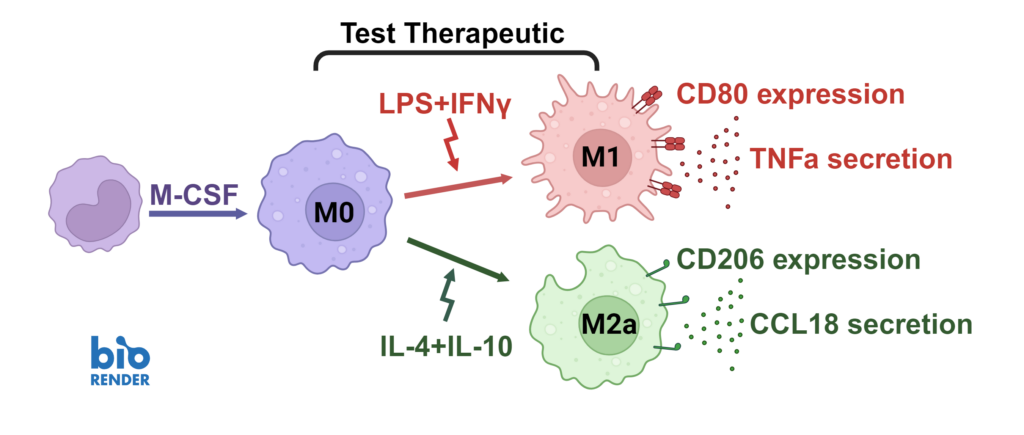
Macrophages were differentiated from monocytes and polarised to M1 or M2a phenotypes following the schema to the left. Polarisation was assessed through CD80 staining and TNFα secretion for M1, and CD206 staining and CCL18 secretion for M2a; staining quantified via HCS analysis. Inhibition of M1 and M2a polarisation was demonstrated using dexamethasone and tofacitinib as reference compounds. Novel therapeutics can be benchmarked against these references.
A THP-1 macrophage cytokine release assay (CRA) can be used to explore modulators of an inflammatory response or “de-risk” any potential unwanted effects of novel therapeutic delivery systems.
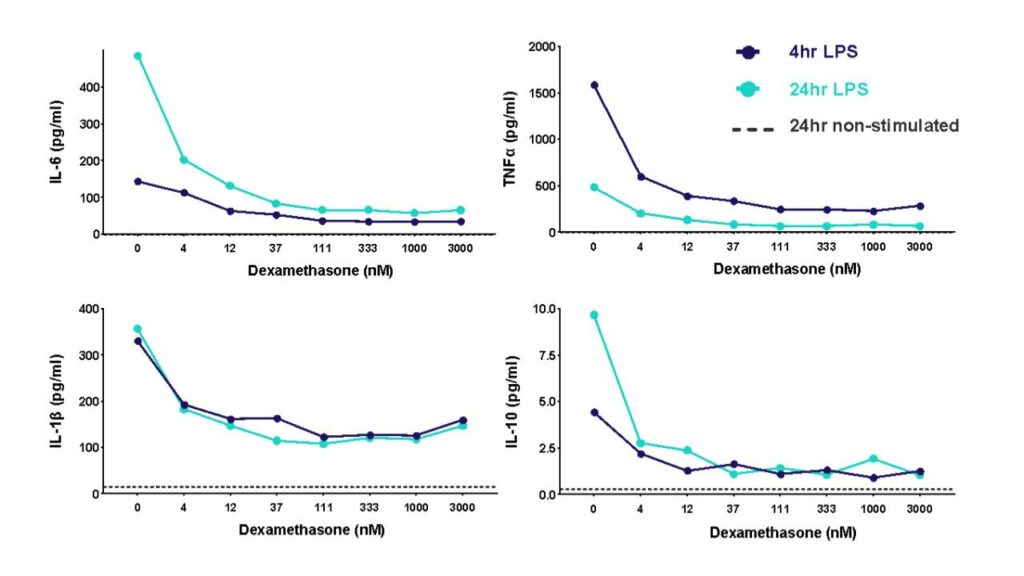
THP-1 cells were differentiated with PMA and then stimulated with LPS to induce cytokine release in the presence of different concentrations of the corticosteroid dexamethasone. Cytokine release was measured at 4h and 24h post LPS stimulation.
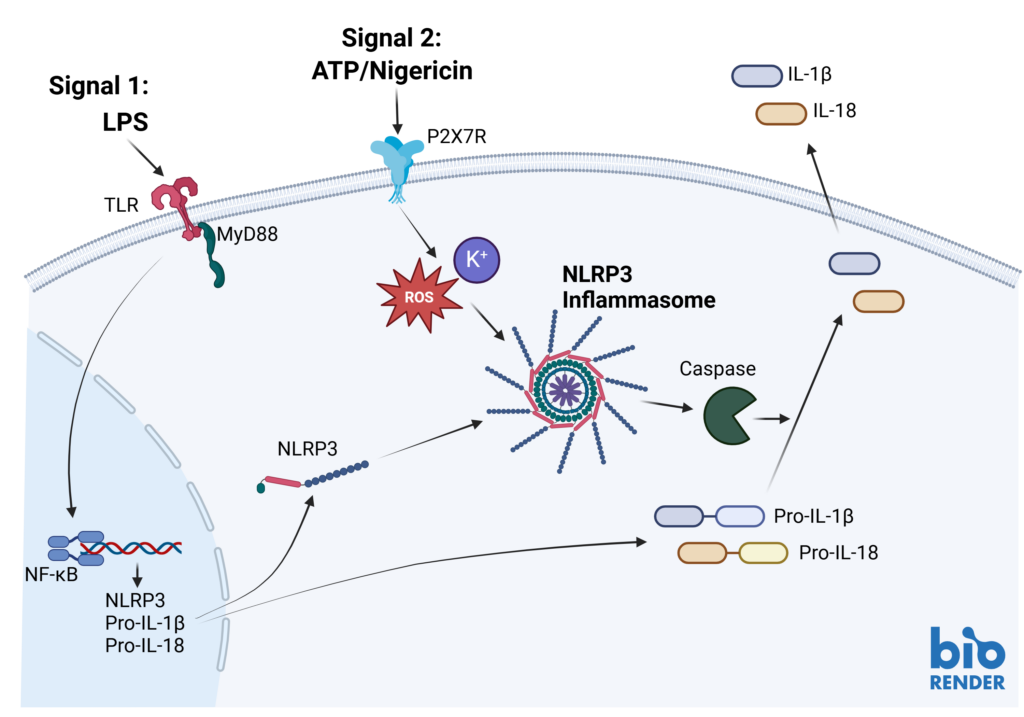
Inflammasome complexes form in response to infection, tissue damage or metabolic imbalances. Once formed the inflammasomes activate Caspase 1 which in turn activated the pro-inflammatory cytokines IL-1β and IL-18. Targeting the inflammasome and the resulting signaling pathways is a useful tool in modulating the immune system.


Effect of MCC950 on Inflammasome activation – IL-1β and IL-18. PBMC from three healthy donors were pretreated with MCC950, vehicle (DMSO) or media for 1 hours prior to stimulation with LPS for 4 hours. Nigericin was then added for a further 45 minutes prior to harvest of the cell culture supernatant for quantification of (A) IL-1β and (B) IL-18 by TR-FRET and ELISA respectively. Data points show mean +/- SEM of technical replicates. One-way ANOVA with Dunnett’s multiple comparisons test comparing stimulated (media) to unstimulated, LPS alone, vehicle and MCC950 to vehicle; *<0.05, **<0.01, ***<0.001.
Other formats: PBMC, macrophage subsets, microglia
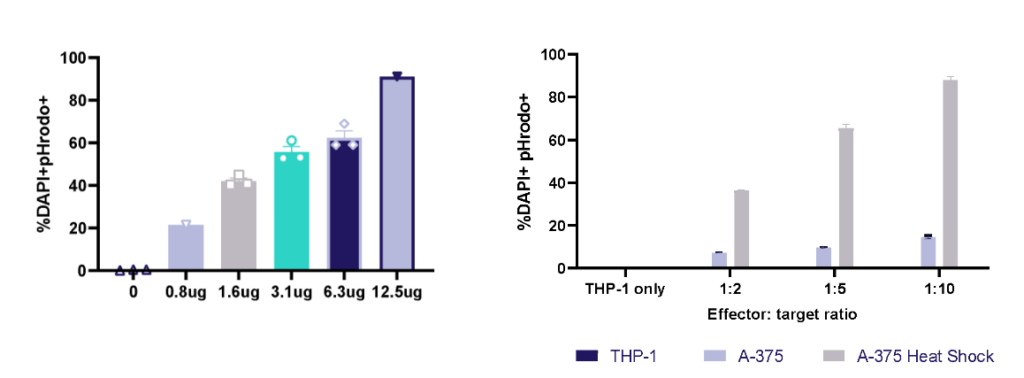
THP-1 macrophage phagocytosis of E.Coli bioparticles and efferocytosis of Heat Shock treated A-375 melanoma tumour cells. THP-1 cells were cultured with E.Coli bioparticles 0-12.5ug/ml; at 1h phagocytosis was evaluated by pHrodo+ cells using flow cytometry. THP-1 cells were cultured with three E:T ratios of A-375 or heat shock treated A-375; at 1h efferocytosis was evaluated by pHrodo+ cells using flow cytometry.
Neutrophil
Inflammation Assays
- Respiratory Burst (ROS, Phagoburst)
- NETs
- Phagocytosis/trogocytosis
Assays
Neutrophils are armed with a variety of effector mechanisms, they release neutrophil extracellular traps (NETs), comprising of chromatin and antimicrobial proteins which are released via a unique, pro-inflammatory form of cell death called NETosis. Dysregulated NET release can damage the host, contributing to autoimmune diseases such as Systemic Lupus Erythematosus (SLE) by the release of autoantigens. Similarly, dysregulated NET release contributes to diseases such as atherosclerosis, deep vein thrombosis and has been shown to promote cancer progression and metastasis. Modulating neutrophil function is therefore a potential therapeutic intervention.
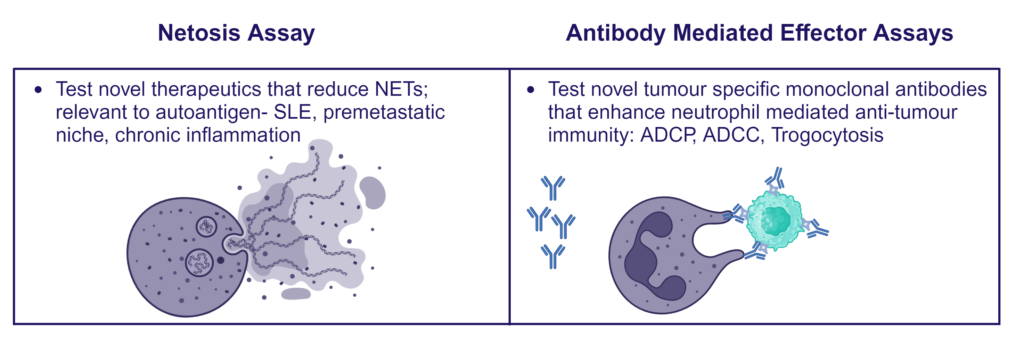
Immune complex-driven NETosis in autoimmune inflammation

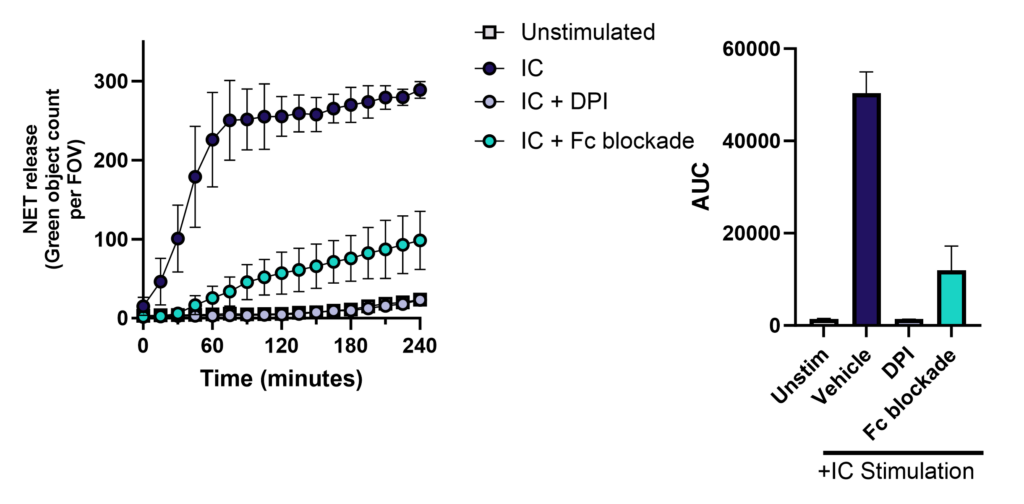
Neutrophils were isolated from fresh blood, cultured with or without NOX2 inhibitor (DPI) or Fc block, and then stimulated with immune complexes (IC) in the presence of SYTOX green to detect NETs. Cells were imaged for green fluorescence (NETs) every 15 minutes for 4 hours. Data shows that NETosis increases in response to stimulation with IC and is inhibited by Fc blockade or DPI.
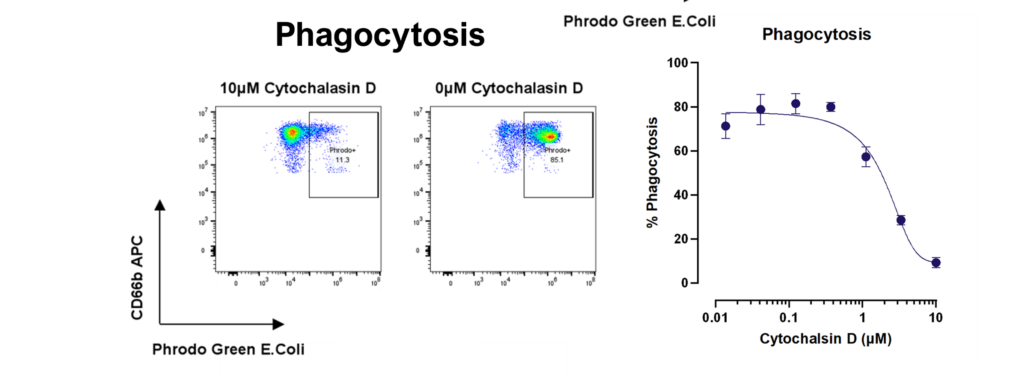
Phagocytosis – Neutrophils were cultured with pHrodo labelled E. coli in the presence or absence of a titration of cytochalasin D and phagocytosis measured by flow cytometry. Phagocytic neutrophils were identified as CD66b+ pHrodo+.
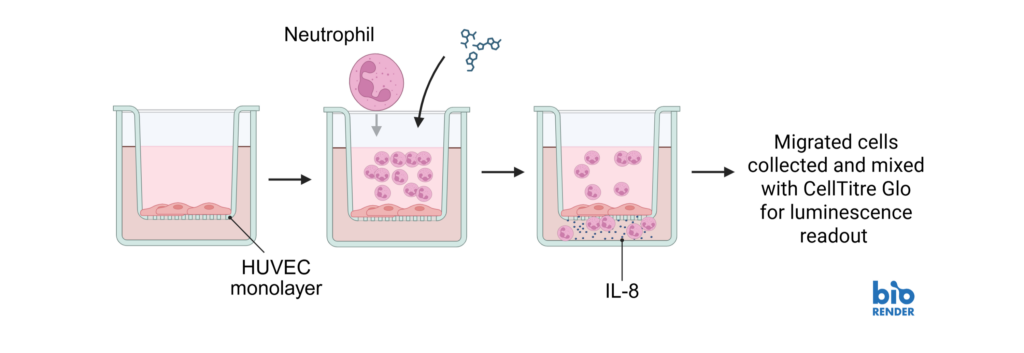
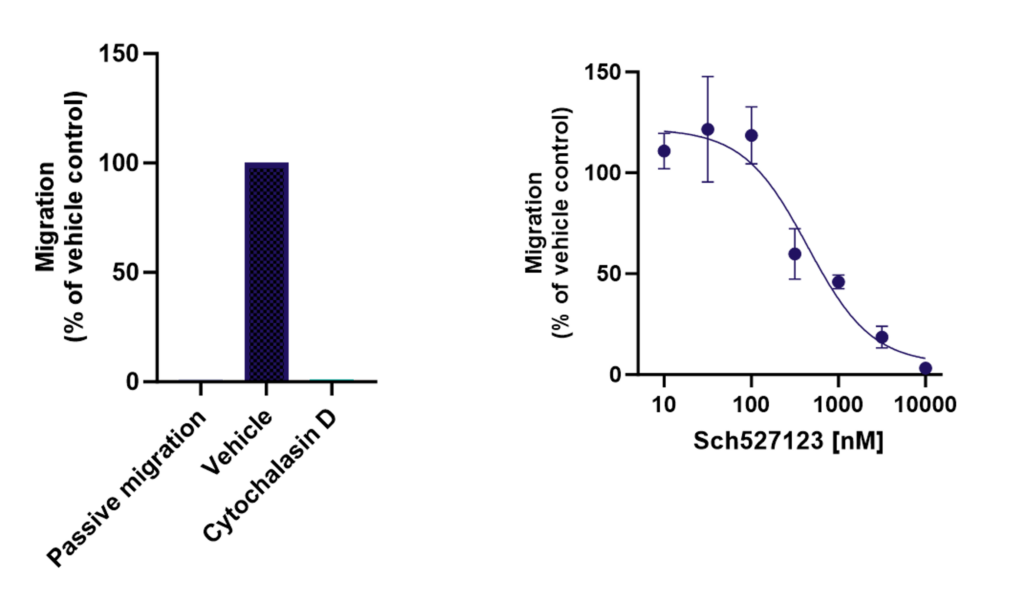
Endothelial cells (HUVEC) were cultured on transwell inserts and confluency confirmed. Neutrophils were isolated from fresh blood and plated on top of the endothelial layer in the presence or absence of CXCR1/2 antagonist (Sch527123), Cytochalasin D, or controls. Chemoattractant IL-8 was added to the lower chamber of the transwell and cells were allowed to migrate before being analysed by luminescence. Sch527123 demonstrated a dose responsive inhibition of neutrophil migration across the endothelial monolayer. Neutrophil migration was inhibited by the positive control, Cytochalasin D.
CD4+ T cell
Immune-Oncology Assays
- Polyclonal
- MLR
- Antigen specificity
- nTreg suppression assay
- iTreg polarisation assay
Autoimmunity Assays
- Th1/2/17 polarisation
- Th17 function
- Tr1/iTreg polarisation
- nTreg suppression
- Tfh, naïve
- CM and EM phenotype/function
- Polyclonal T cell proliferation
- Cytokine release
Inflammation Assays
- Cytokine release
Assays
ELISpot is a highly sensitive method to test immune modulators, novel vaccine candidates or de-risk immunogenicity testing in an antigen-specific CD4 and/or CD8 T cell assay.
| Condition | Aim |
| No Stimulation | Negative control |
| PMA | Positive control |
| CERI (CMV, EBV, RSV, Influenza) | MHC-I restricted peptide pool to evaluate modulation of CD8+ T-cell memory response |
| CPI (CMV, Parainfluenza, Influenza) | Positive protein antigens to evaluate modulation of CD4+ T-cell memory response |
| CEF (CMV, EBV, Influenza) | MHC-I restricted peptide pool to evaluate modulation of CD8+ T-cell memory response |
| Cyclosporin A (CsA) | Inhibition of immune response |
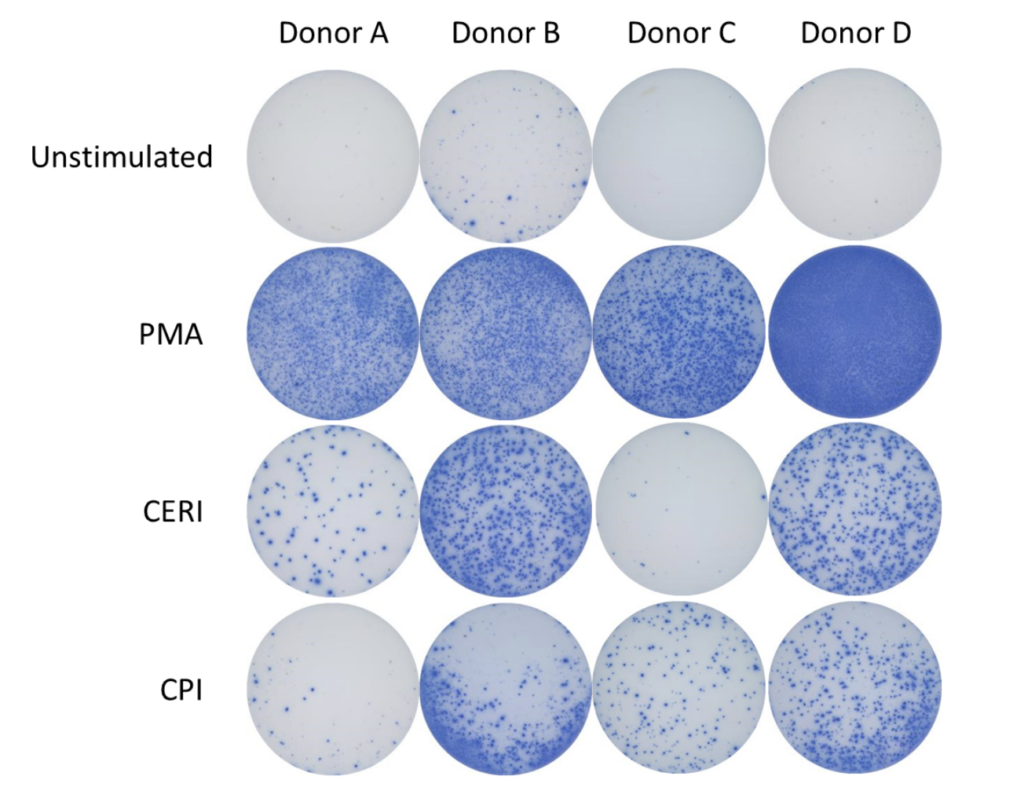
Figure 1: The ELISpot visual image results showing four donors under four conditions: Unstimulated (negative control), stimulated with PMA (positive control), stimulated with CERI, and stimulated with CPI antigens. Each purple spot represents one IFNγ producing cell.
Spot Forming Units (SPU) for IFN-γ per 100,000 PBMC from CERI, CPI and CEF antigens for three donors
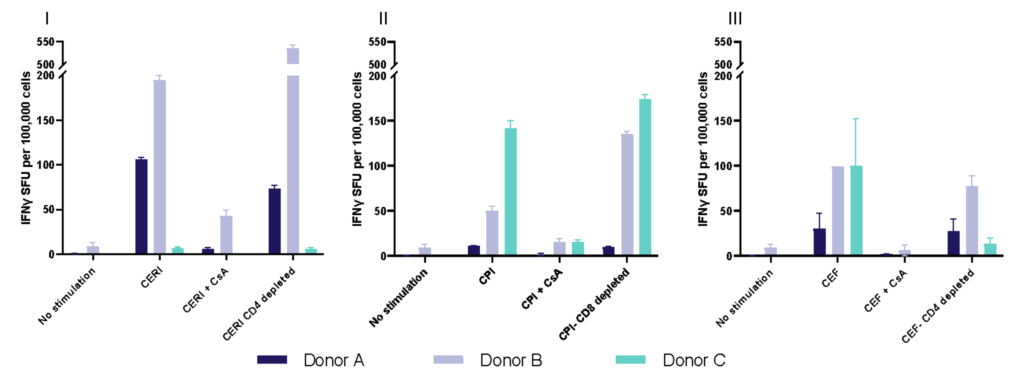
Figure 2: T cell responses from three donors using stimuli, CERI (I), CPI (II) and CEF (III). Expressed as spot forming units per 100,000 donor PBMC. Depletion of CD4 or CD8 T cells demonstrates specificity of response to peptide pools. Each value represents the mean of triplicate wells ±SEM.
T helper 17 (Th17) cells, are a subset of CD4 T cells distinguished by their production of IL-17. While crucial for protecting the body against extracellular pathogens, Th17 cells have also been implicated in autoimmune disease. Discovering therapies that target either the differentiation or effector function of Th17 cells could therefore prove beneficial for the treatment of some autoimmune diseases.
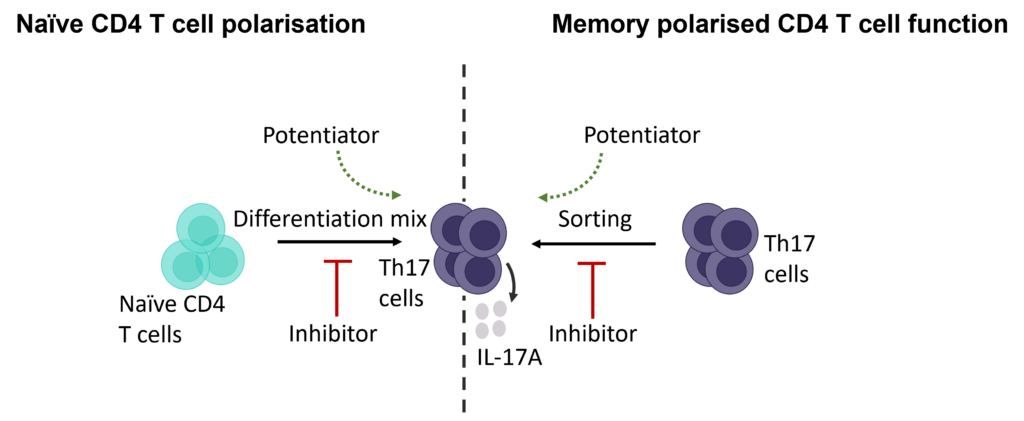
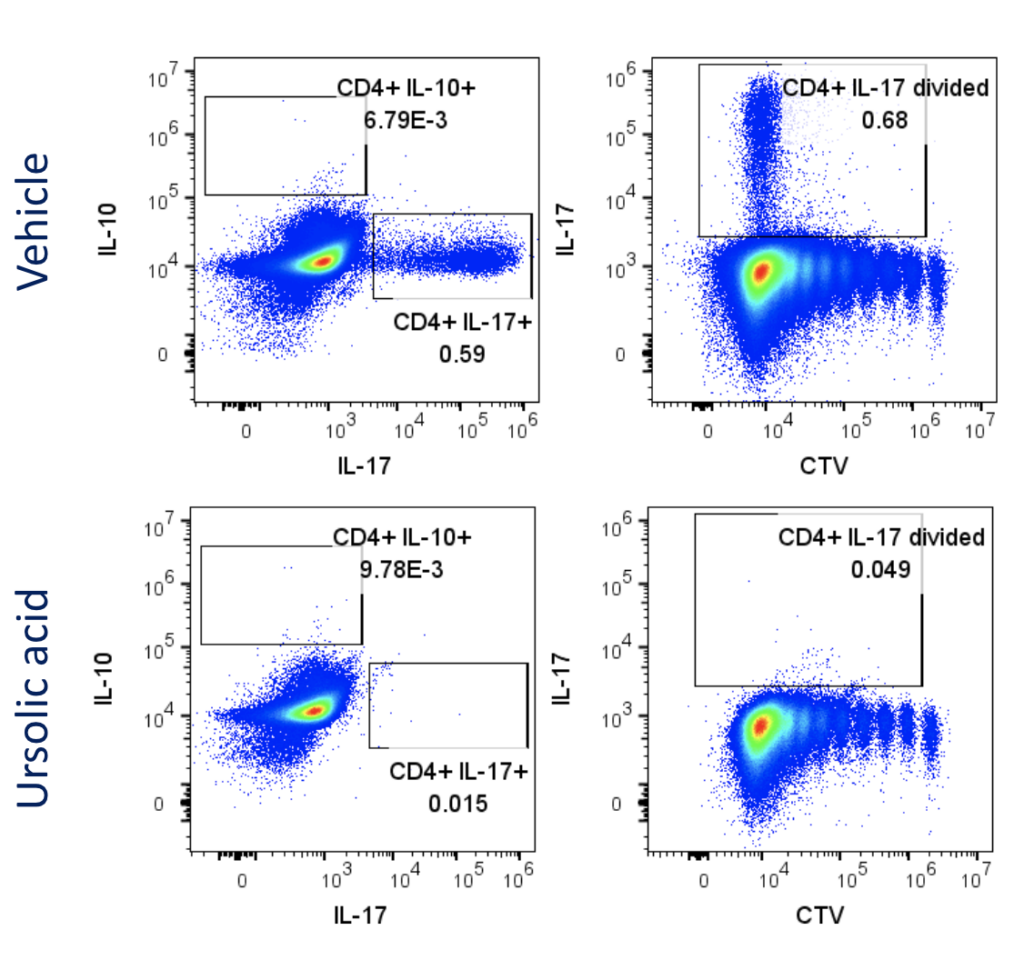
Evaluation of therapeutic modulation of Th17 differentiation
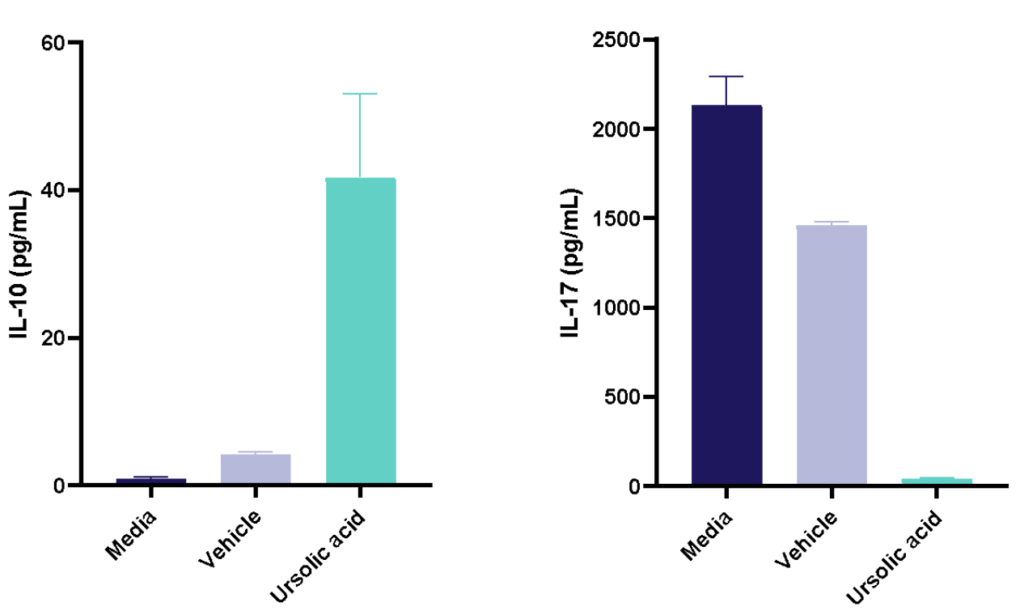
Figure 1: Polarisation of Th17 cells. Naïve CD4 cells were cultured under Th17 polarising conditions for 12 days in the presence or absence of Ursolic Acid. CD4 T cells were assessed for proliferation by CTV dilution; intracellular cytokine staining (ICS) of IL-17 and IL-10 by flow cytometry. On Day 12, Supernatants were collected and evaluated for IL-17 and IL-10 levels by MSD. RoRγT inhibitor Ursolic Acid showed selective inhibition of IL-17 production by intracellular cytokine staining (ICS) and MSD.
Evaluation of therapeutic modulation of Th17 effector function
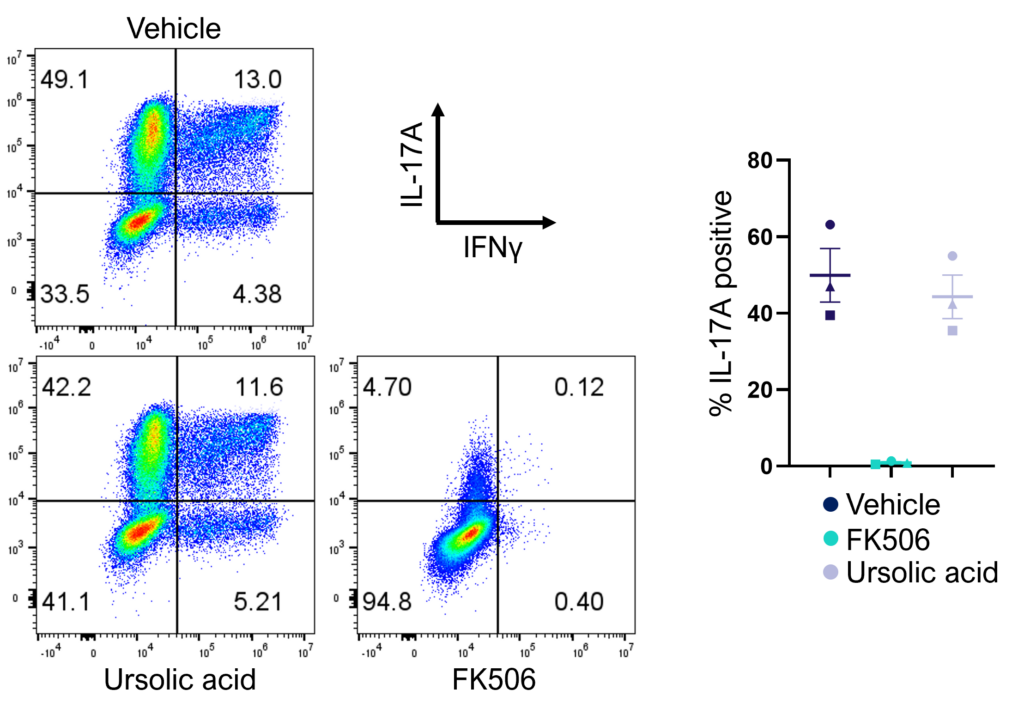
Figure 2: Inhibition of Th17 cell effector function. Magnetically sorted memory Th17 cells were polyclonally stimulated under Th17 conditions in the presence or absence of ursolic acid or FK506 for 5 days. A percentage of Th17 cells are polyfunctional (IL- 17A+IFNγ+) and refractory to inhibition by ursolic acid (an inhibitor of Th17 differentiation) but not FK506.
For many the humble “workhorse” mixed lymphocyte reaction (MLR) is a key screening assay for immuno-therapeutics. 1-way (DC and T cells) and 2-way MLRs (PBMC) provide expression of key ligands on human immune cells for therapeutic modulation.
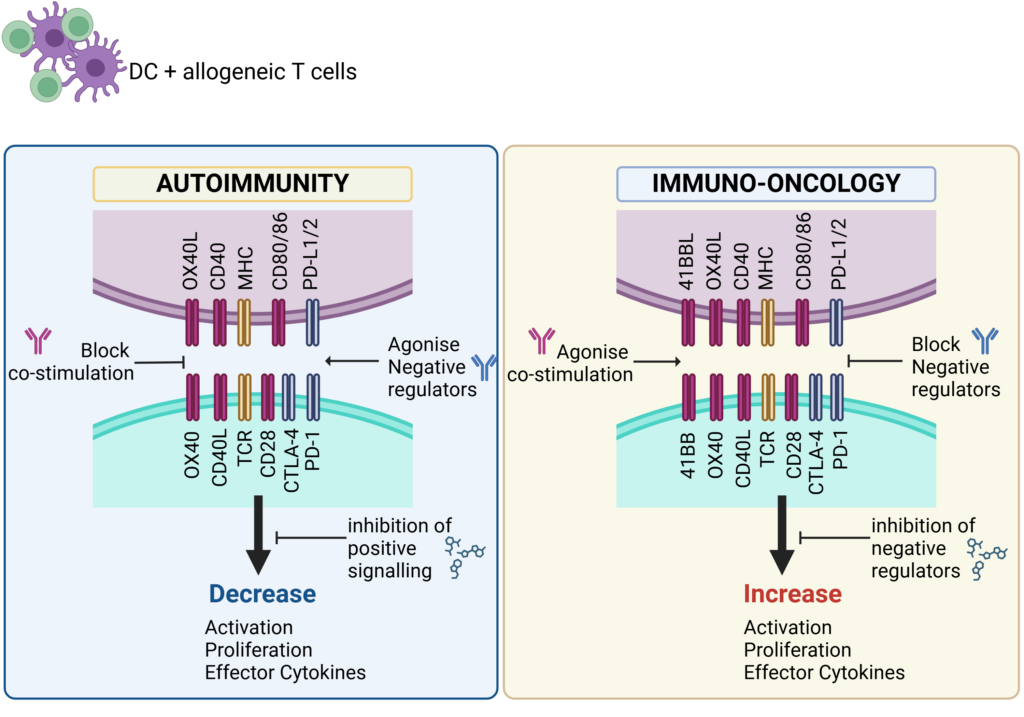
Schematic showing key therapeutic targets that are blocked (antagonise) or engaged (agonise) to control immune responses in Autoimmunity or boost immune responses for Immuno-Oncology.
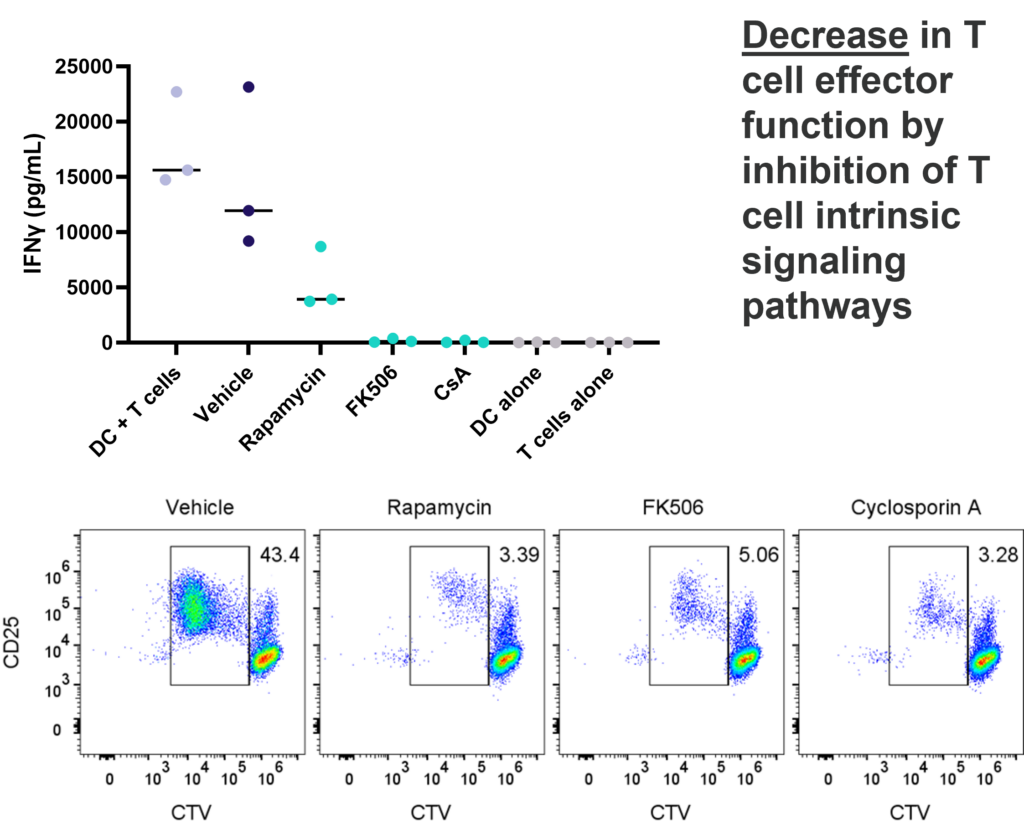

1-way or 2-way MLR. One way MLR of CTV labelled T cells in response to allogenic DC, matured with LPS. T cell proliferation and IFNγ production is inhibited by Rapamycin, FK506 and Cyclosporin A, in contrast, PD-1 blockade (Pembrolizumab) increases T cell IFNγ production, but does not enhance T cell proliferation.
This assay provides a relevant human system to test novel therapeutics that block or engage key co-stimulatory or inhibitory ligands expressed on immune cells.
Natural Tregs (nTregs) are a thymus-derived subset of CD4+ T cells found in peripheral blood, capable of suppressing immune responses to maintain tolerance and homeostasis. Suppression assays model Treg function by measuring their ability to inhibit effector T cell proliferation, activation, and cytokine production. Altering Treg suppressive function can have promising therapeutic potential, both in the context of autoimmunity and immuno-oncology.
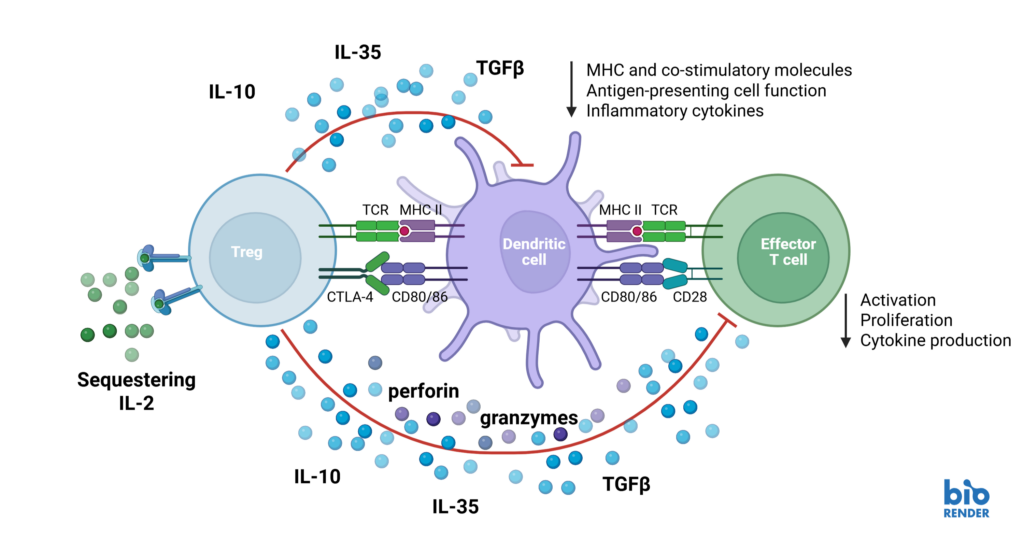
Schematic showing mechanisms by which regulatory T cells suppress effector T cell responses.
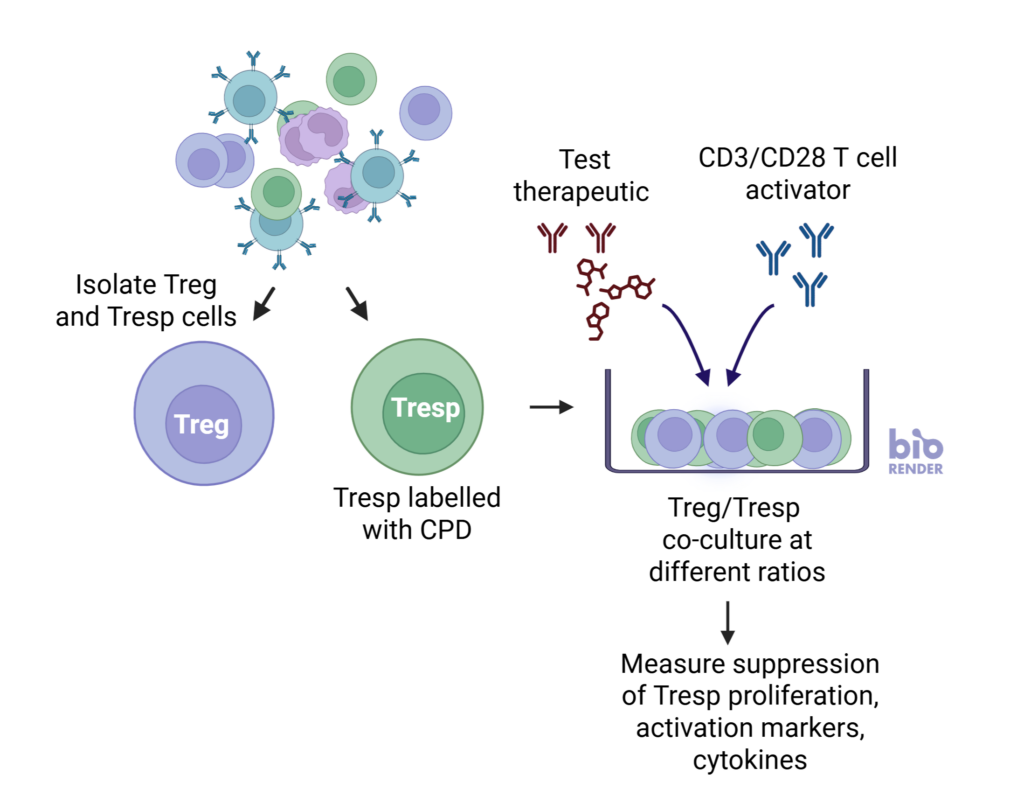
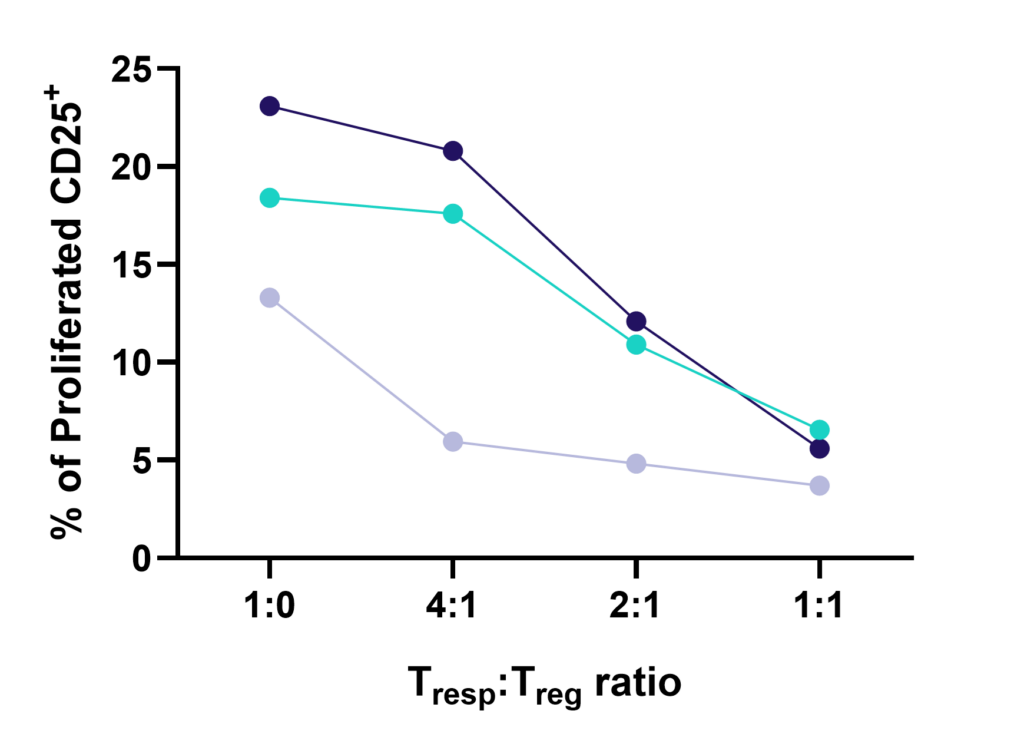
nTreg cells suppress the proliferation of Tresp cells in a ratio-dependent manner
nTregs were co-cultured with stimulated CD3 T cells at three Tresp:Treg ratios. Graphs show increased suppression of Tresponder proliferation in the presence of increasing number of Tregs. Data shown from three healthy donors.
I. T cell response to Tetanus Toxoid, Influenza and PPD antigens
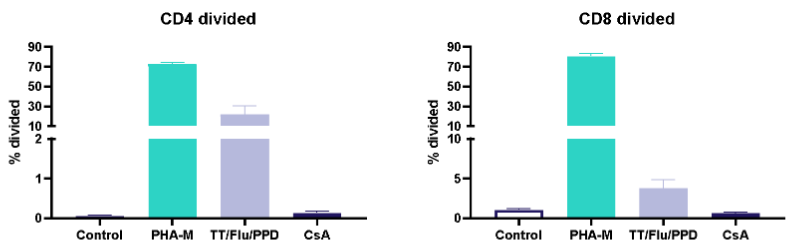
II. Dose response to Influenza antigen
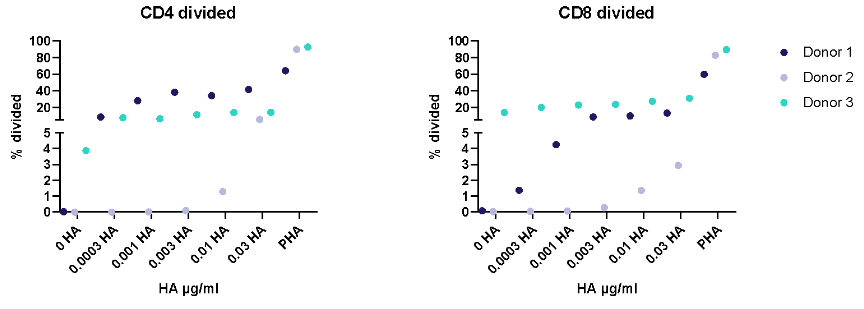
Antigen specific T cells response to a recall antigens. (I) Healthy donors PBMC were stimulated with PHA-M or triple antigen cocktail (Tetanus Toxoid, Influenza and PPD). Cyclosporin was used as a reference treatment. (II) Dose response to Influenza antigen. CD4 and CD8 T cells proliferation was measured by flow cytometry using CTV dilutions.
Monocyte
Inflammation Assays
Assays
Inflammasomes are key signaling platforms within the immune system. Inflammasome complexes form in response to infection, tissue damage or metabolic imbalances. Once formed the inflammasomes activate Caspase 1 which in turn activated the pro-inflammatory cytokines IL-1β and IL-18. Targeting the inflammasome and the resulting signaling pathways is a useful tool in modulating the immune system.



Effect of MCC950 on Inflammasome activation – IL-1β and IL-18.
PBMC from three healthy donors were pretreated with MCC950, vehicle (DMSO) or media for 1 hours prior to stimulation with LPS for 4 hours. Nigericin was then added for a further 45 minutes prior to harvest of the cell culture supernatant for quantification of (A) IL-1β and (B) IL-18 by TR-FRET and ELISA respectively. Data points show mean +/- SEM of technical replicates. One-way ANOVA with Dunnett’s multiple comparisons test comparing stimulated (media) to unstimulated, LPS alone, vehicle and MCC950 to vehicle; *<0.05, **<0.01, ***<0.001.
Other formats: PBMC, macrophage subsets, microglia
Cytokine release assay can be used to “de-risk” any potential unwanted effects of novel biotherapeutics in accordance with FDA and EMA recommended guidelines.
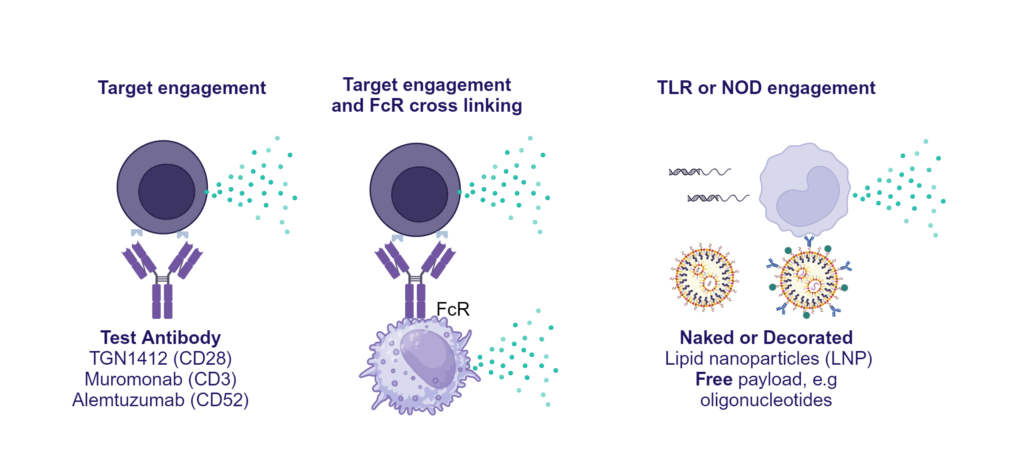
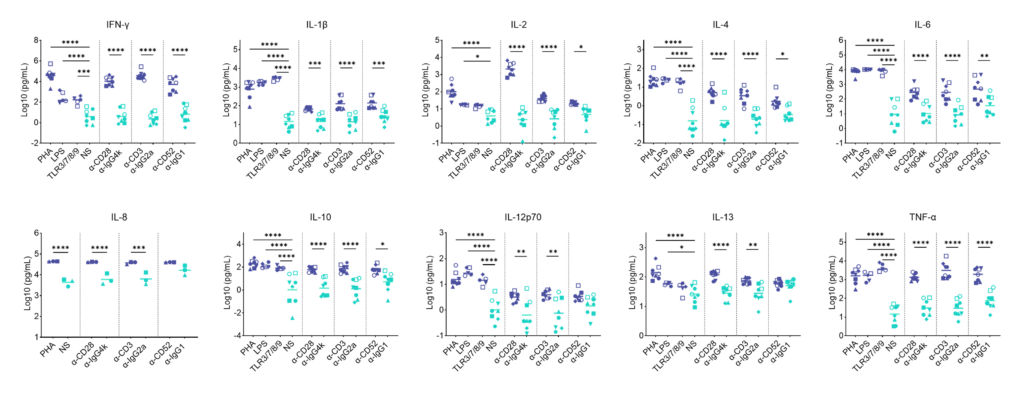
Preclinical safety PBMC cytokine release assay (CRA)
PBMC solid phase CRA: release of IFN-γ, IL-1β, IL-2, IL-4, IL-6, IL-8, IL-10, IL-12p70, IL-13 and TNF-α in response to a panel of antibodies coated onto plastic or TLR stimuli or polyclonal T cell stimuli (PHA).
PBMC isolated from buffy coats were added to wells coated with a-CD28 (TGN1412), a-CD3 (Muromonab), a-CD52 (Alemtuzumab) or with isotype control antibodies, or were stimulated with PHA, LPS, or a cocktail of TLR3/7/8/9 agonists. Cytokine release was measured at 48hr post stimulation. NS = Non-stimulated; Graphs show log10 transformed data with mean of 10 donors indicated. One way ANOVA with Sidak’s multiple comparisons test performed on log10 transformed data with significance indicted as *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001. Note for IL-8 where values were above the ULOD these were assigned ULOD value.
Other readouts: frozen disease PBMC
Other formats: Whole Blood CRA
Other immuno-tox assays: include ADCC/ CDC/ ADCP
Preclinical safety whole blood cytokine release assay (CRA)

Whole blood soluble CRA; release of IFN-γ, IL-1β, IL-2, IL-4, IL-6, IL-8, IL-10, IL-12p70, IL-13 and TNF-α in response to a panel of antibodies or polyclonal T cell stimuli (PHA).
Whole blood was added to wells containing a-CD28 (TGN1412), a-CD3 (Muromonab), a-CD52 (Alemtuzumab) or with isotype control antibodies or were stimulated with PHA. Cytokine release was measured at 48hr post stimulation by MSD. NS = Not stimulated. Graphs show log10 transformed data with mean of 10 donors indicated. One way ANOVA with Sidak’s multiple comparisons test performed on log10 transformed data with significance indicted as *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001. Note for IL-8 where values were above the ULOD these were assigned the ULOD value.
Eosinophil
Inflammation Assays
- Contact us for further details.
Fibroblast
Immune-Oncology Assays
- CAF-multicellular tumour killing assays
Inflammation Assays
- FMT
Assays
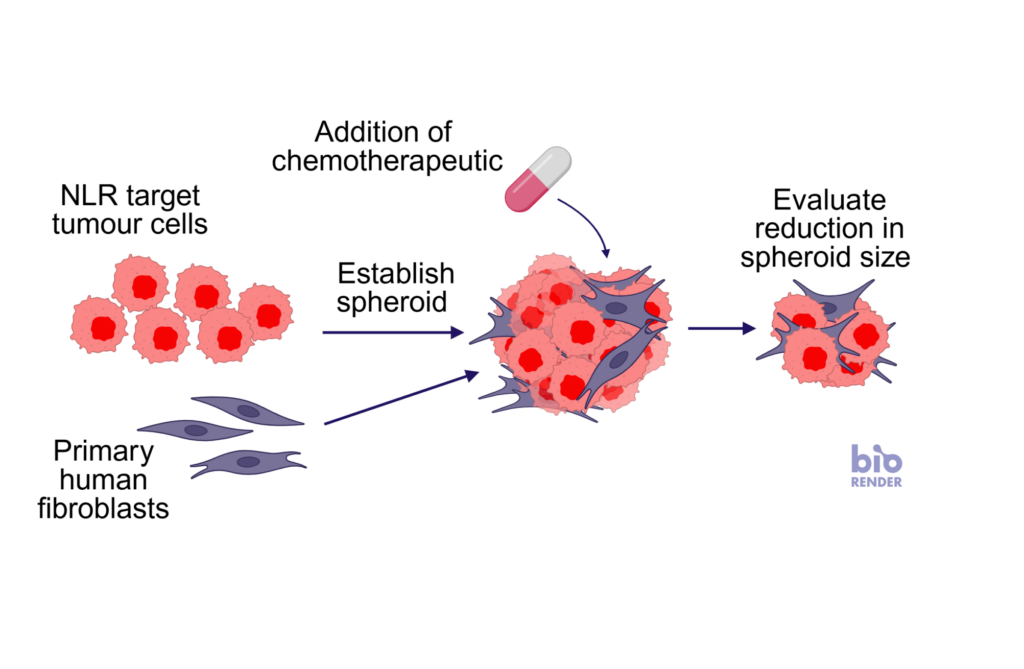
Tumour cells initiate cancer and drive disease progression, however many other cell types surrounding the tumour, including stromal and immune cells promote carcinogenesis. Cancer associated fibroblasts (CAFs) play a critical role in supporting tumour growth, shaping the cell microenvironment and promoting therapeutic resistance. Co-culture of tumour cells with fibroblasts has been shown to create a more organotypic tumour-like environment, enabling more physiologically relevant responses for therapeutic testing.
Formation of fibroblast-tumour spheroids
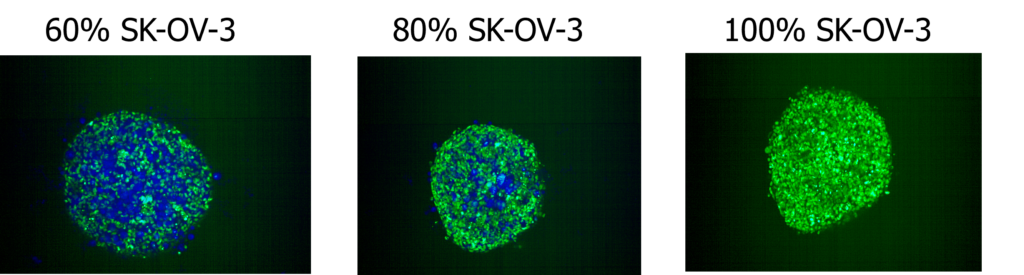
SK-OV-3/Fibroblast Spheroid Formation at 48-hours. SK-OV-3 NLR and CellTrace™ Blue-labelled NHDF cells were seeded into ultra low attachment 96-well plates and allowed to form spheroids over 72-hours. Images were acquired on the Opera Phenix High Content Screening System, 10x magnification. Images displayed above are from representative wells with indicated SKOV-3 and NHDF cell compositions.
SK-OV-3/Fibroblast Spheroid killing following Cisplatin treatment
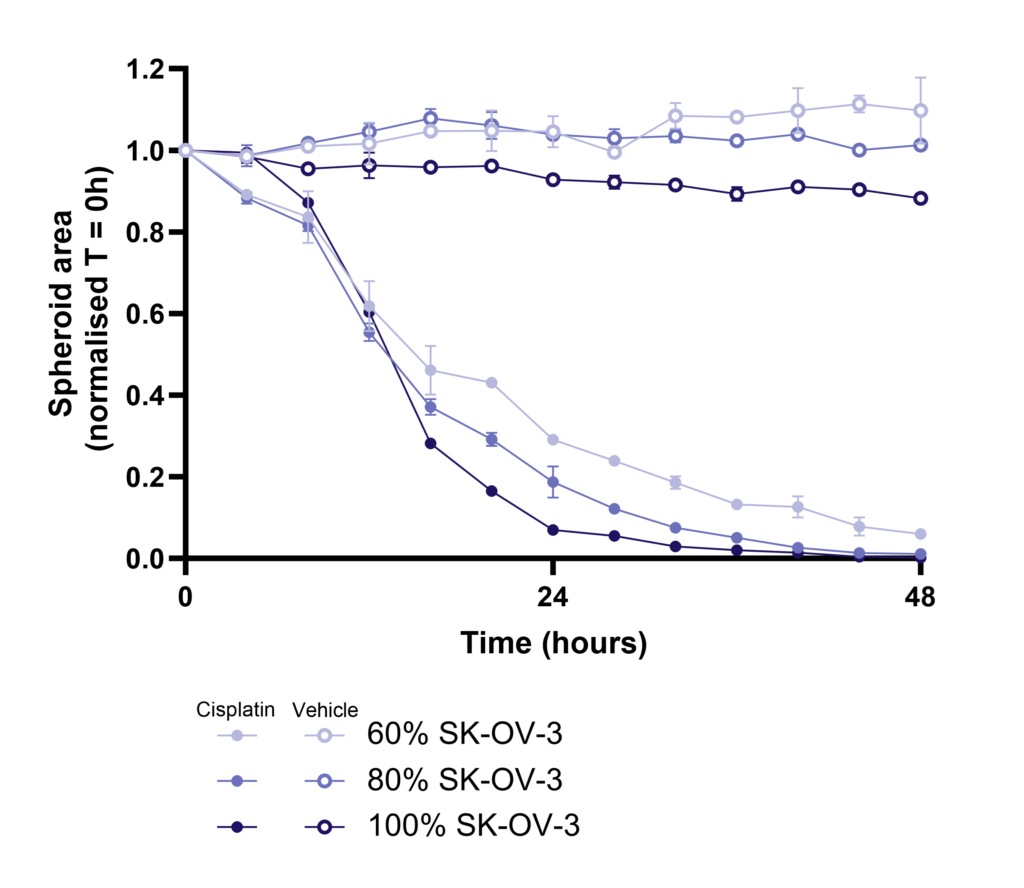
Time course of SK-OV-3/Fibroblast Spheroid killing following Cisplatin treatment. Spheroids were generated over 96-hours with a total of 5,000 cells per spheroid. Spheroids were treated with Cisplatin (100µM), vehicle or media control with varying percentages of SKOV-3 and NHDF cells (50-100% SKOV-3 cells) making up the spheroid. TKA was performed with a 1:1 ratio of PBMC: Spheroid. Images were acquired every 4-hours for 72-hours. Images were acquired on the Opera Phenix High Content Screening System, 10x magnification.
Epithelial Cell
Immune-Oncology Assays
- On/off target assay
- EMT
Inflammation Assays
- EMT
- Keratinocyte cytokine release
- Epithelial interface-barrier integrity
- Tight junction
- TEER
- Cytokine release; Epithelial/immune cell co-culture
Mast Cell
Inflammation Assays
- Contact us for further details.
Basophil
Inflammation Assays
- Contact us for further details.
Innate Lymphoid Cell
Autoimmunity Assays
- ILC1/2/3 phenotype
- Cytokine production
Inflammation Assays
- ILC1/2/3 phenotype a
- Cytokine production
